- Blog
- Nestle milo
- Magicprefs yosemite
- Play diner dash hometown hero playfirst
- Game of thrones beyond the wall cast
- The count of monte cristo book cover
- Fica quieta mical cifra
- Nursery pal cloud baby monitor
- Mortal kombat 11 pc
- Doomsday defense
- Mario kart wii download for usb loader
- Gamemaker studio 2 forums
- Features of google earth pro
- 3947 net monitor
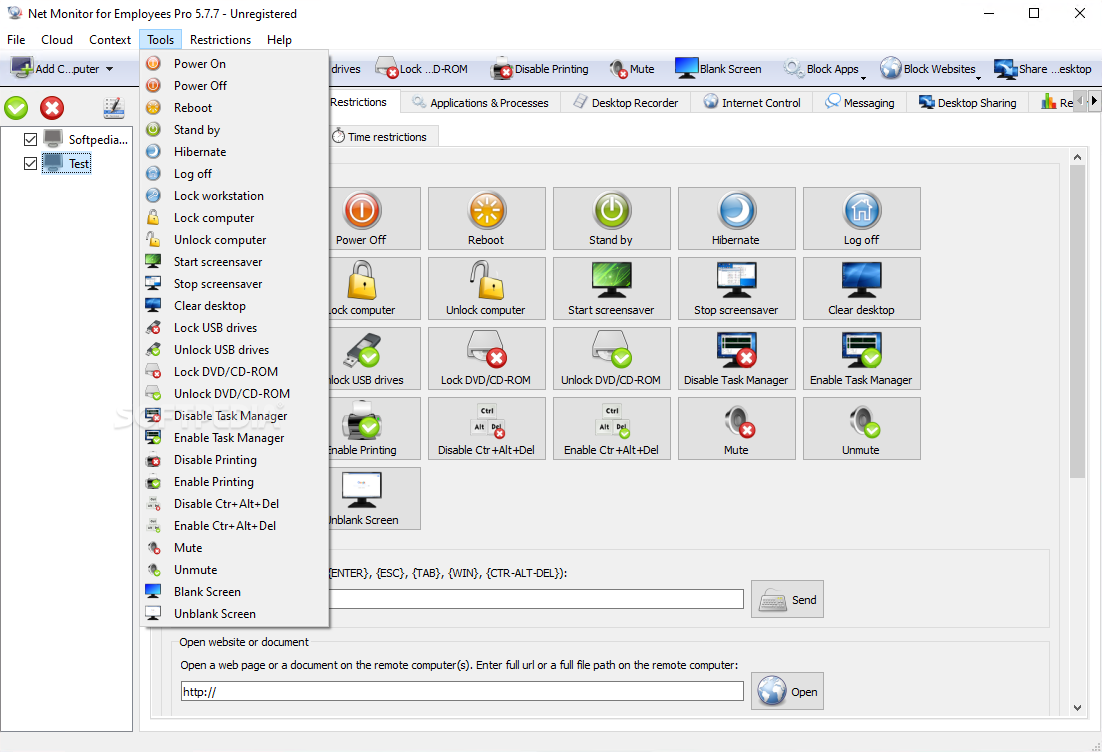
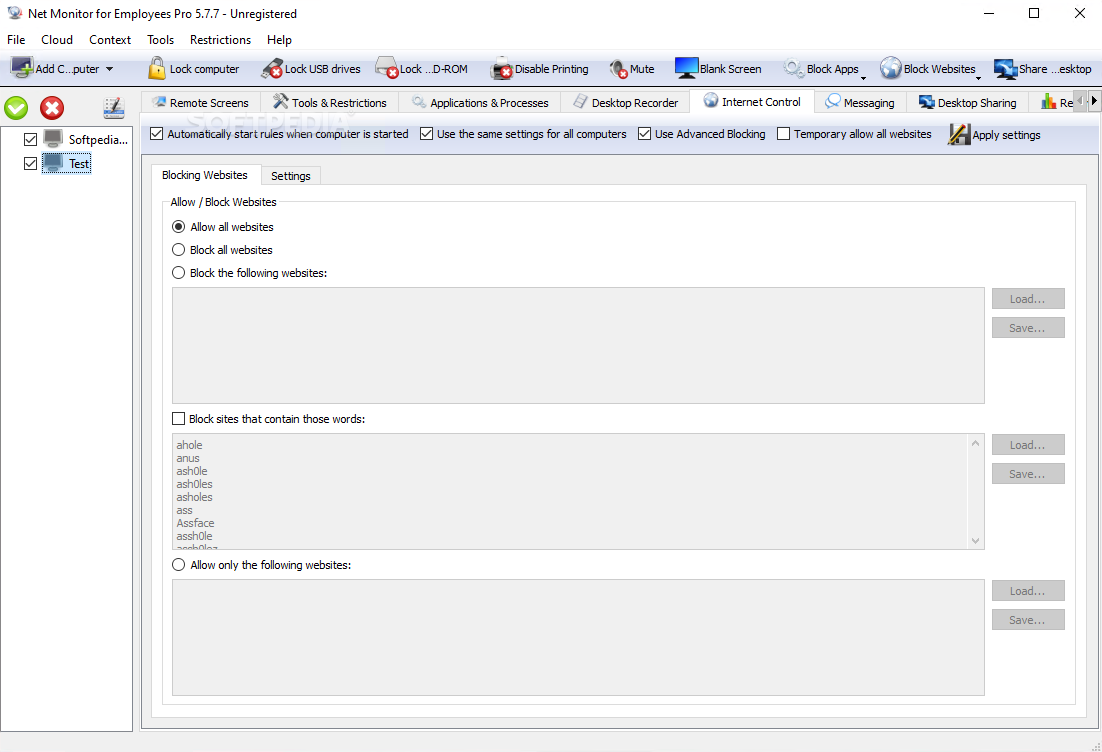
#3947 net monitor serial
Because cervical lesions may take longer to manifest in a young population, 4vHPV vaccine type (6, 11, 16, and 18) persistent infection (defined by the detection of HPV by polymerase chain reaction in serial sampling over a specified duration) was assessed. Anal and oropharyngeal end points were not included. Analyses for effectiveness included assessment for genital warts and cervical and genital precancers and cancers as well as persistent infection. Effectiveness was assessed only in the follow-up period. The base study evaluated immunogenicity only. These rates were compared with the observed incidence rates in previous efficacy studies within the 4vHPV vaccine program at Merck & Company Inc. Instead, the effectiveness of vaccination with the 4vHPV vaccine was assessed by calculating the incidence of disease end points (EVG and CVG cohorts). 4 However, the vaccine must enable a sustained immune response and long-term effectiveness to protect individuals because HPV acquisition risks are greater later in life.īecause there were no placebo groups, vaccine efficacy measurements could not be assessed.
#3947 net monitor series
3 When administered between 11 and 12 years old, the HPV vaccination series can be initiated along with the meningococcal and tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis, absorbed vaccines. Earlier vaccination produces more robust antibody responses, 2 and the impact of early vaccination (in grades 8 and 9 at ∼13 years old) in reducing cervical dysplasia and genital warts is already evident ∼4 years later.
1 Although approved for both boys and girls who are 9 to 26 years old, initiating the vaccination series before exposure to HPV is important.
This prophylactic vaccine prevents cervical, vulvar, vaginal, and anal cancer caused by HPV types 16 and 18, genital warts caused by HPV types 6 and 11, and precancerous cervical, vulvar, vaginal, and anal lesions caused by HPV types 6, 11, 16, and 18. Ten years have passed since the licensure of Gardasil (4-valent human papillomavirus vaccine, recombinant ) in the United States. POTENTIAL CONFLICT OF INTEREST: Dr Ferris has received grant support from Merck & Company Inc (Kenilworth, NJ) through his institution and personal fees for consultancy and advisory boards for Merck & Company Drs Block and Lazcano-Ponce have received research grants from and are members of a speaker’s bureau for Merck & Company and have served as paid expert witnesses and consultants for Merck & Company Dr Mehlsen has received funding from Merck & Company to conduct human papillomavirus–vaccine clinical trials Dr Chatterjee has received funding from Merck & Company and GlaxoSmithKline to conduct human papillomavirus–vaccine clinical trials and served on a speaker’s bureau for Merck & Company Dr Iversen has received compensation from Merck & Company to conduct vaccine clinical trials and for scientific advisory board fees Drs Joshi, Chu, Krick-Likos, Saah, and Das are employees of Merck Sharp & Dohme Corporation, a subsidiary of Merck & Company and may hold stock options the other authors have indicated they have no potential conflicts of interest to disclose.
- Blog
- Nestle milo
- Magicprefs yosemite
- Play diner dash hometown hero playfirst
- Game of thrones beyond the wall cast
- The count of monte cristo book cover
- Fica quieta mical cifra
- Nursery pal cloud baby monitor
- Mortal kombat 11 pc
- Doomsday defense
- Mario kart wii download for usb loader
- Gamemaker studio 2 forums
- Features of google earth pro
- 3947 net monitor
